|
3/29/2024 0 Comments Modern atomic theory
For example, copper and chlorine can form a green, crystalline solid with a mass ratio of 0.558 g chlorine to 1 g copper, as well as a brown crystalline solid with a mass ratio of 1.116 g chlorine to 1 g copper. The law of multiple proportions states that when two elements react to form more than one compound, a fixed mass of one element will react with masses of the other element in a ratio of small, whole numbers. Previously, an atom was defined as the smallest part of an element that maintains the identity of that element. 4), is a fundamental concept that states that all elements are composed of atoms. (c) In the cathode ray, the beam (shown in yellow) comes from the cathode and is. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. Thomson produced a visible beam in a cathode ray tube. For example, there are many compounds other than isooctane that also have a carbon-to-hydrogen mass ratio of 5.33:1.00.ĭalton also used data from Proust, as well as results from his own experiments, to formulate another interesting law. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 1.5. The results of these measurements indicated that these particles were much lighter than atoms ( Figure 2.2.1 ). (c) In the cathode ray, the beam (shown in yellow) comes from the cathode and is accelerated past the anode toward a fluorescent scale at the end of the tube. 
That is, samples that have the same mass ratio are not necessarily the same substance. Thomson produced a visible beam in a cathode ray tube. It is worth noting that although all samples of a particular compound have the same mass ratio, the converse is not true in general. THE SMALLEST AND LIGHTEST POSITIVE ION WAS OBTAINED FROM HYDROGEN AND WAS CALLED PROTON.\) The behavior of these particles in a magnetic or electric field is opposite to that of electrons or cathode rays. Some positively charged particles carry a multiple of a fundamental unit of electric charge.Ĥ. Learn more about the history, facts, and experiments of the atomic theory. Foundations of Modern Atomic Theory: Thomson, Rutherford, and Bohr Introduction According to Schwab (1974) scientific inquiry tends to look for patterns of change and relationships which constitute the heuristic (explanatory) principles of our knowledge. It was developed by John Dalton and others in the 19th century and has undergone continuous refinement with quantum mechanics and relativity. The charge to mass ratio of particles depends on the gas from which it originates.ģ. The modern atomic theory is the scientific theory of matter based on the fact that the chemical elements are composed of aggregations of similar subunits (atoms) with nuclear and electron substructure. Those are simply the positively charged gaseous ions.Ģ. Previously, we defined an atom as the smallest part of an element that maintains the identity of that element. All matter is made up of tiny particles called atoms. 1 ), is a fundamental concept that states the following: 1. Dalton thought that atoms were the smallest units of matter tiny, hard spheres that could not be broken down any further. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 4.3.1 4.3. However, one of his underlying assumptions was later shown to be incorrect. The positively charged particles depend upon the nature of gas present in the cathode ray tube. Daltons ideas proved foundational to modern atomic theory. The experiment for canal rays was carried out in modified cathode ray tube, by E. the cathode rays consist of electrons, while the anode/canal rays are the positively charged gaseous ions. This model is patterned on the solar system and is known as the planetary model. Each element has its own atomic number, which is equal to the number of protons in its nucleus. 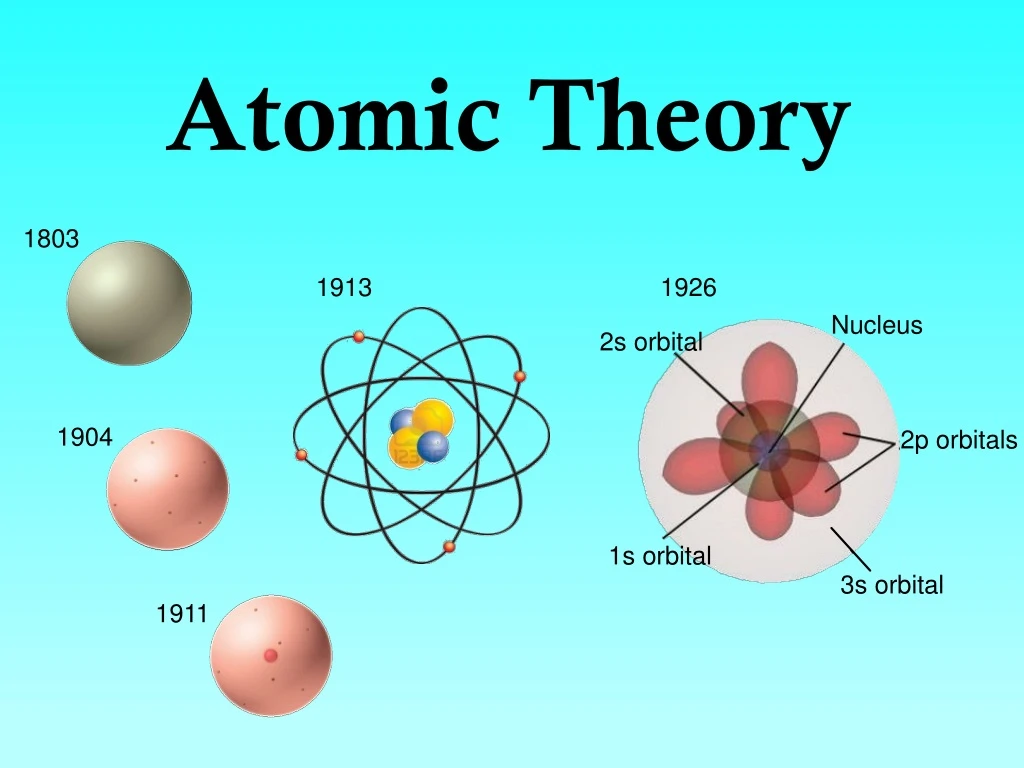
Atoms themselves are composed of protons, neutrons, and electrons. He proposed that electrons are arranged in concentric circular orbits around the nucleus. Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms. You are right, both kinds of rays are emitted simultaneously. In 1913, Neils Bohr, a student of Rutherford s, developed a new model of the atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
